(RxWiki News) The US Food and Drug Administration (FDA) has approved a new gene therapy for vision loss.
The FDA approved Luxturna (voretigene neparvovec-rzyl) to treat adults and children who have an inherited form of vision loss that can lead to blindness.
This new medication is the first directly administered gene therapy in the United States to be approved to treat retinal dystrophy caused by mutations in a specific gene. And in the case of Luxturna, the specific mutation is the biallelic RPE65 mutation.
“Today’s approval marks another first in the field of gene therapy — both in how the therapy works and in expanding the use of gene therapy beyond the treatment of cancer to the treatment of vision loss — and this milestone reinforces the potential of this breakthrough approach in treating a wide range of challenging diseases," said FDA Commissioner Dr. Scott Gottlieb in a press release.
The type of retinal dystrophy Luxturna treats is rare and affects approximately 1,000 to 2,000 patients across the US. Those with the biallelic RPE65 mutation will experience vision loss that gets worse over time and will ultimately progress to complete blindness.
“Patients with biallelic RPE65 mutation-associated retinal dystrophy now have a chance for improved vision, where little hope previously existed,” said Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, in a press release.
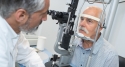
A surgeon must inject Luxturna into a specific area of the eye. Luxturna treatment will be surgically performed in each eye on separate days with at least six days between procedures. Common side effects include eye redness, cataracts, increased intraocular pressure and retinal tearing.
The FDA granted approval of Luxturna to Spark Therapeutics Inc.
Written By Anyssa Garza, PharmD, BCMAS